 If a stimulus induces a change in the balance of fission and fusion in a cell, it could significantly alter the mitochondrial network. The processes of fission and fusion oppose each other and allow the mitochondrial network to constantly remodel itself. Definitively, fission is the event of a single entity breaking apart, whereas fusion is the event of two or more entities joining to form a whole. Mitochondria are highly versatile and are able to change their shape through fission and fusion events. As researchers learn more about these proteins and how they reach the respective mitochondrial compartments that utilize them, it becomes evident that there is a multitude of processes that work together in the cell to allow for mitochondrial biogenesis. During the past two decades, researchers have discovered over thirty proteins that participate in mitochondrial protein import. Other proteins may have targeting information in their sequences and will not include an N-terminal signal. Some proteins will have an N-terminal targeting signal, and these proteins will be detected and transported into the matrix, where they will then be cleaved and folded. All proteins will enter the mitochondria by a translocase on the outer mitochondrial membrane (TOM). Precursor proteins will be transported to one of four areas of the mitochondria, which include the outer membrane, inner membrane, intermembrane space, and matrix. The resulting unfolded precursor proteins will then be able to reach their respective mitochondrial compartments. First, mRNA is translated in the cell’s cytosol. Since the majority of mitochondrial protein comes from the nuclear genome, the proteins need to be properly targeted and transported into the mitochondria to perform their functions. Mitochondrial proteins encoded from the nuclear genome need to be targeted and transported appropriately into the mitochondria. Critically, mitochondrial numbers and morphology vary according to cell type and context-specific demand, whereby the balance between mitochondrial fusion/fission regulates mitochondrial distribution, morphology, and function. However, depending on the energy substrates available and the redox state of the cell, the cell may increase or decrease the number and size of mitochondria. Mitochondrial biogenesis increases metabolic enzymes for glycolysis, oxidative phosphorylation and ultimately a greater mitochondrial metabolic capacity. The majority of mitochondrial protein comes from the nuclear genome, while the mitochondrial genome encodes parts of the electron transport chain along with mitochondrial rRNA and tRNA. Mitochondria are produced from the transcription and translation of genes both in the nuclear genome and in the mitochondrial genome. It is postulated that higher mitochondrial copy number (or higher mitochondrial mass) is protective for the cell. The mitochondrion is a key regulator of the metabolic activity of the cell, and is also an important organelle in both production and degradation of free radicals. 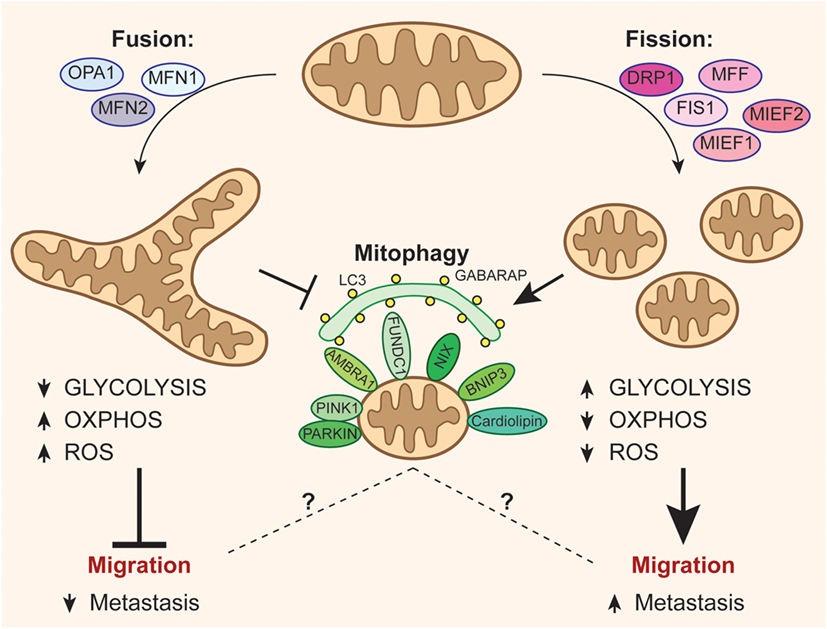
However, recent evidence suggests that mitochondria may have evolved without symbiosis. It is commonly thought that mitochondria descend from cells that formed endosymbiotic relationships with α-protobacteria they have their own genome for replication. The ability for a mitochondrion to self-replicate is rooted in its evolutionary history. Mitochondrial biogenesis is activated by numerous different signals during times of cellular stress or in response to environmental stimuli, such as aerobic exercise. It was first described by John Holloszy in the 1960s, when it was discovered that physical endurance training induced higher mitochondrial content levels, leading to greater glucose uptake by muscles. Mitochondrial biogenesis is the process by which cells increase mitochondrial numbers. Process by which cells build mitochondrial mass 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
